Ethan's BGGN 213 Portfolio
Class work for bioinformatics
class07
Ethan Ashley (PID: A15939817) 2025-10-22
K-means Clustering
Today we will begin exploring some “classical” machinge learning approaches. We will start with clustering:
Let’s first make up some data to cluster where we know what the answer should be.
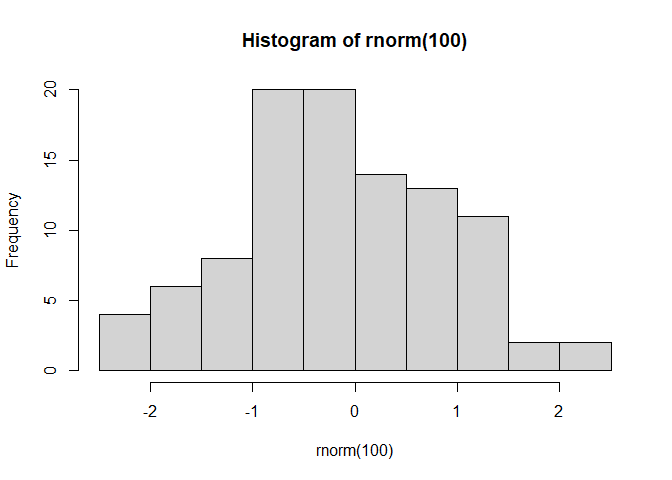
#rnorm randomly picks n values around a normal distribution, unless specified it assumes mean = 0 and SD = 1
hist(rnorm(100))
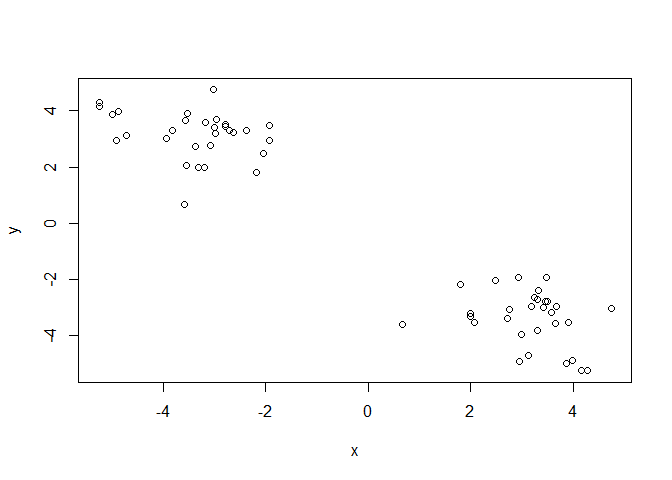
Let’s generate 30 points centered around +3 and -3
x <- c(rnorm(30, 3), rnorm(30, -3))
#y <- c(rnorm(30, 3), rnorm(30, -3))
y <- rev(x)
#can be used to compare two vectors side by side column-wise
z <- cbind(x,y)
#visual inspection of our randomly generated data
plot(z)
The main function in “base” R for K-means clustering is called
kmeans().
#kmeans(matrix of data, centers = number of clusters to make)
k <- kmeans(z, 2)
k$cluster #returns the cluster vector of which points are assigned to which cluster
## [1] 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 2 2 2 2 2 2 2 2
## [39] 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2
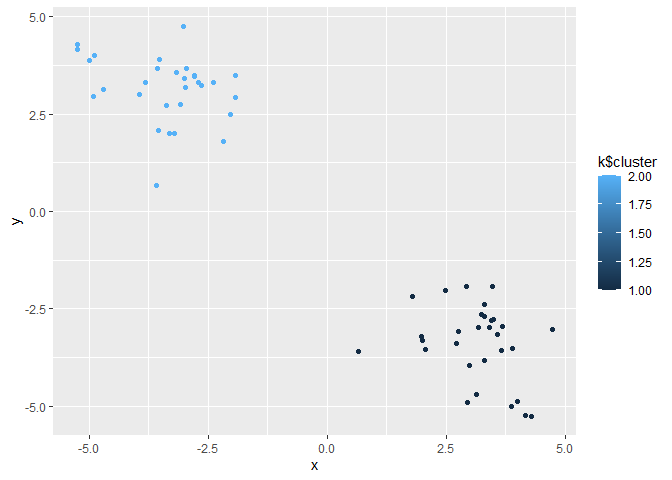
Q. Make a plot of our data colored by cluster assignment?
library(ggplot2)
ggplot(z) + aes(x, y, color=k$cluster) + geom_point()
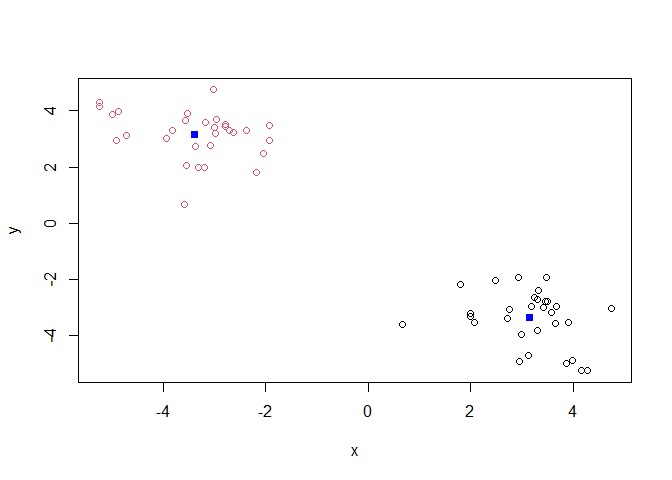
plot(z, col=k$cluster)
points(k$centers, col="blue", pch=15)
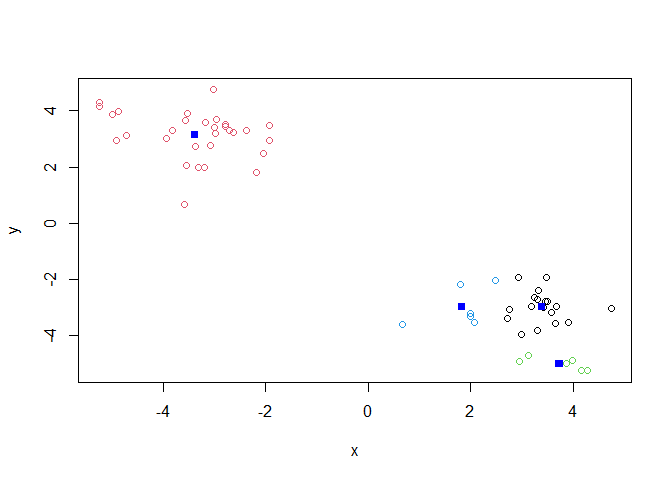
Q. Cluster with k-means into 4 clusters and plot your results as above.
k4 <- kmeans(z, 4)
plot(z, col=k4$cluster)
points(k4$centers, col="blue", pch=15)
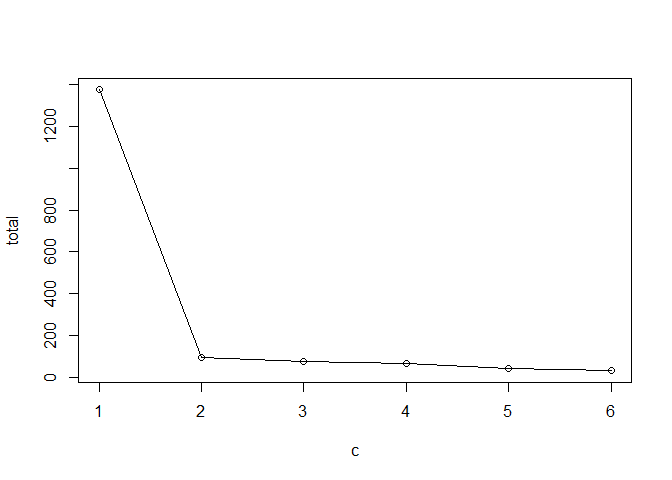
Q. Run k-means with centers (i.e. values of k) from 1 to 6. Make a plot of the tot.withinss vs. cluster number
# store this value for each analysis k$tot.withinss
c <- 1:6
total <- NULL
for (i in c) {
total <- c(total, kmeans(z, i)$tot.withinss)
}
plot(c, total, typ="o") #called a "Scree Plot", used to determine ideal # of groups with K-means clustering
This method imposes a structure on the data.
Hierarchical Clustering
The main funciton in “base” R for this is called hclust(). It peforms
bottom-up clustering, iteratively generating fewer clusters each time.
#uses a distance matrix instead of raw data like K-means clustering
#must apply dist() function to raw data
d <- dist(z)
hc <- hclust(d)
hc
##
## Call:
## hclust(d = d)
##
## Cluster method : complete
## Distance : euclidean
## Number of objects: 60
#produces a clustering "tree"
plot(hc)
abline(h=7, col="red")
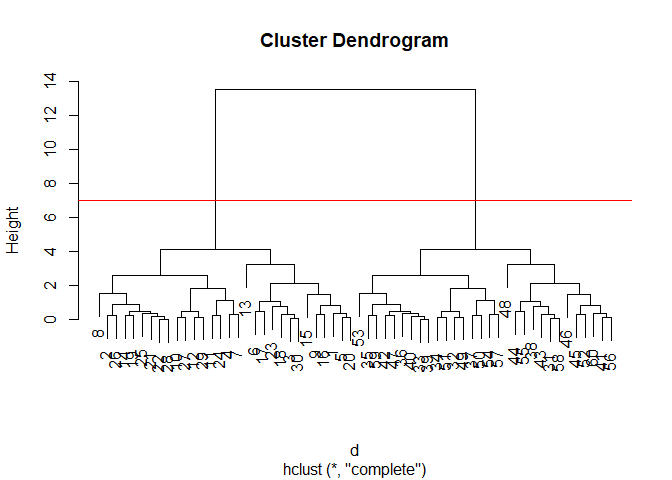 Important
information in the dendrogram is conveyed by the location of the
horizontal bars.
Important
information in the dendrogram is conveyed by the location of the
horizontal bars.
To obtain clusters from our hclust result object hc we “cut” the
tree to yield different sub branches. For this we use the cutree()
function. This function yields a clustering vector like with k-means
clustering.
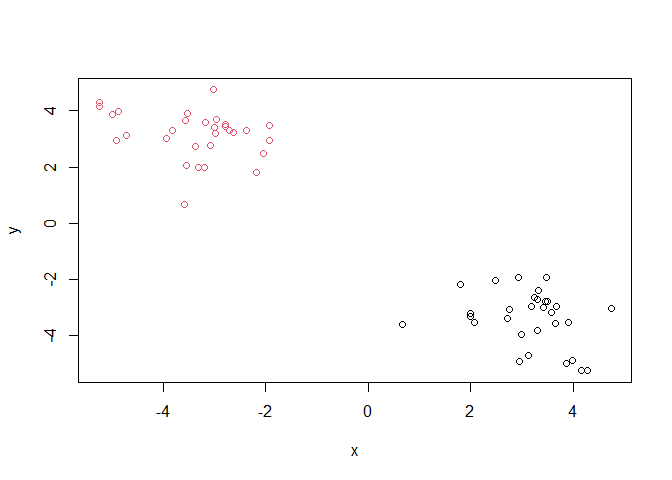
grps <- cutree(hc, k = 2)
plot(z, col=grps)
Principal Component Analysis (PCA)
Example data analysis on UK food data.
url <- "https://tinyurl.com/UK-foods"
x <- read.csv(url)
Q1. How many rows and columns are in your new data frame named x? What R functions could you use to answer this questions?
dim(x)
## [1] 17 5
There are 17 rows and 5 columns in this dataset.
Fixing the row names issue
# Note how the minus indexing works
rownames(x) <- x[,1]
x <- x[,-1]
head(x)
## England Wales Scotland N.Ireland
## Cheese 105 103 103 66
## Carcass_meat 245 227 242 267
## Other_meat 685 803 750 586
## Fish 147 160 122 93
## Fats_and_oils 193 235 184 209
## Sugars 156 175 147 139
Q2. Which approach to solving the ‘row-names problem’ mentioned above do you prefer and why? Is one approach more robust than another under certain circumstances?
I think that specifying the row-names column while importing the data is a preferable way to solve this issue. I think if the structure of the data is known, which it is in this case, importing it in the form we wish to work with it will be more efficient than importing the data and re-formating afterwards. This is also more robust because you can avoid accidentally delting additional columns from the matrix when the chunk is run again.
x <- read.csv(url, row.names=1)
head(x)
## England Wales Scotland N.Ireland
## Cheese 105 103 103 66
## Carcass_meat 245 227 242 267
## Other_meat 685 803 750 586
## Fish 147 160 122 93
## Fats_and_oils 193 235 184 209
## Sugars 156 175 147 139
# Using base R
barplot(as.matrix(x), beside=T, col=rainbow(nrow(x)))
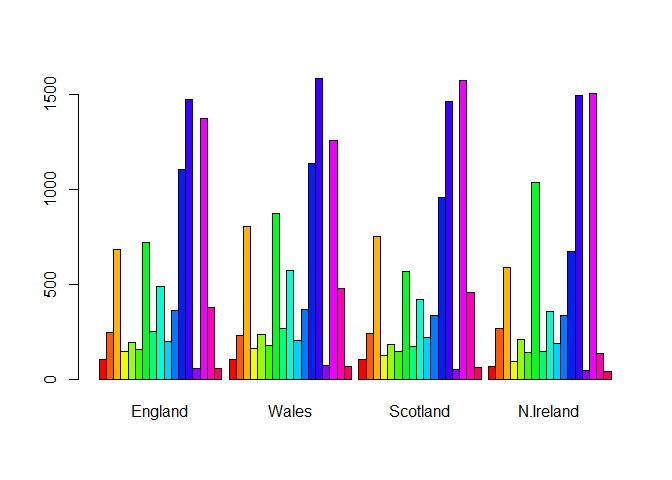
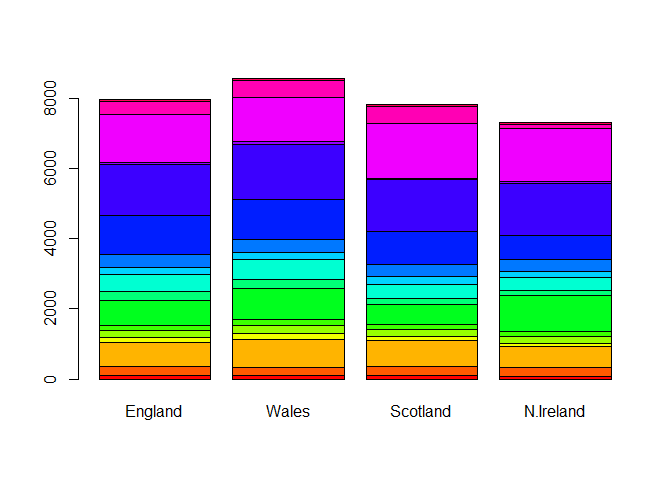
Q3. Changing what optional argument in the above barplot() function results in the following plot?
Changing the value of beside from true to false changes the orientation of our barplot.
# Using base R
barplot(as.matrix(x), beside=F, col=rainbow(nrow(x)))
Reformatting our data to be optmizied for ggplot.
library(tidyr)
# Convert data to long format for ggplot with `pivot_longer()`
x_long <- x |>
tibble::rownames_to_column("Food") |>
pivot_longer(cols = -Food,
names_to = "Country",
values_to = "Consumption")
dim(x_long)
## [1] 68 3
Making a plot with ggplot of the re-organized data.
# Create grouped bar plot
library(ggplot2)
ggplot(x_long) +
aes(x = Country, y = Consumption, fill = Food) +
geom_col(position = "dodge") +
theme_bw()
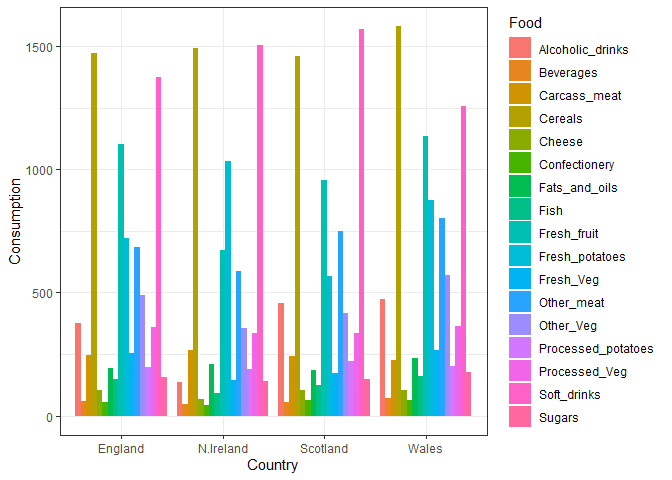
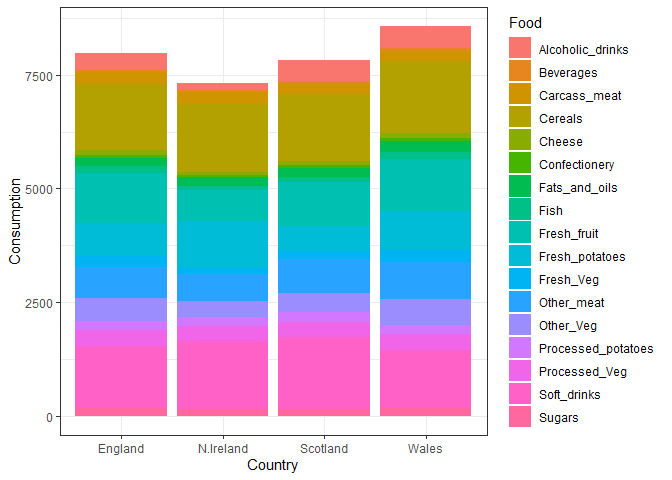
Q4. Changing what optional argument in the above ggplot() code results in a stacked barplot figure?
Changing the position input in geom_col to “stack” changes the
orientaiton of the bar plot
ggplot(x_long) +
aes(x = Country, y = Consumption, fill = Food) +
geom_col(position = "stack") +
theme_bw()
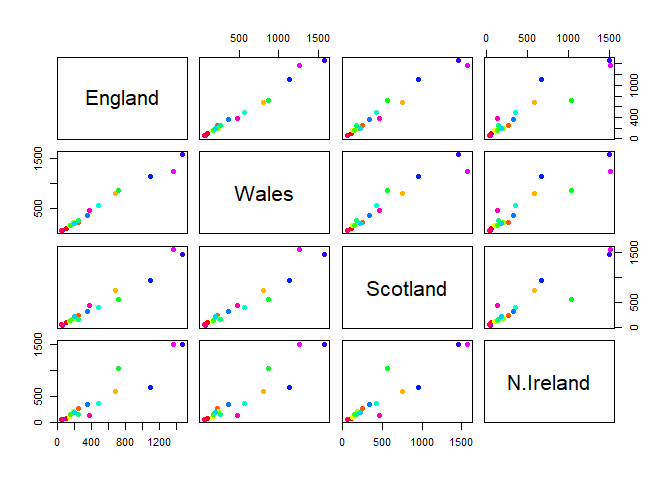
Q5. We can use the pairs() function to generate all pairwise plots for our countries. Can you make sense of the following code and resulting figure? What does it mean if a given point lies on the diagonal for a given plot?
It shows how similar the consumption of different food items is between two countries. Points on the diagonal line are more similar to each other, while those farther away are less similar (i.e., one is greater than another).
pairs(x, col=rainbow(nrow(x)), pch=16)
library(pheatmap)
pheatmap( as.matrix(x) )
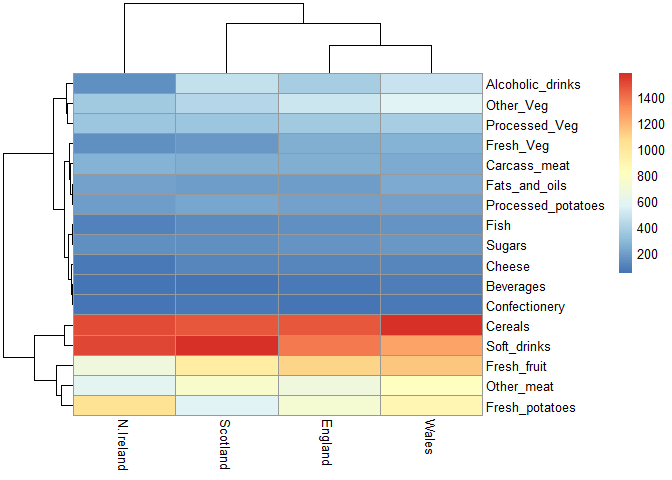
Q6. Based on the pairs and heatmap figures, which countries cluster together and what does this suggest about their food consumption patterns? Can you easily tell what the main differences between N. Ireland and the other countries of the UK in terms of this data-set?
England, Wales, and Scotland appear to cluster together while Northern Ireland is in its own cluster.
# Use the prcomp() PCA function
pca <- prcomp( t(x) )
summary(pca)
## Importance of components:
## PC1 PC2 PC3 PC4
## Standard deviation 324.1502 212.7478 73.87622 3.176e-14
## Proportion of Variance 0.6744 0.2905 0.03503 0.000e+00
## Cumulative Proportion 0.6744 0.9650 1.00000 1.000e+00
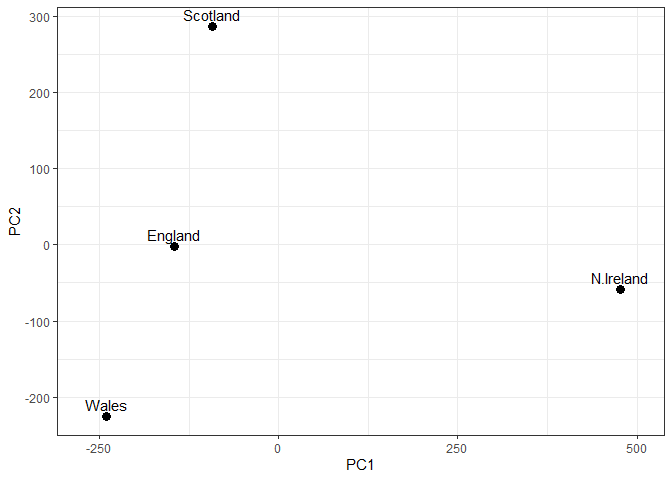
Q7. Complete the code below to generate a plot of PC1 vs PC2. The second line adds text labels over the data points.
# Create a data frame for plotting
df <- as.data.frame(pca$x)
df$Country <- rownames(df)
# Plot PC1 vs PC2 with ggplot
ggplot(pca$x) +
aes(x = PC1, y = PC2, label = rownames(pca$x)) +
geom_point(size = 3) +
geom_text(vjust = -0.5) +
xlim(-270, 500) +
xlab("PC1") +
ylab("PC2") +
theme_bw()
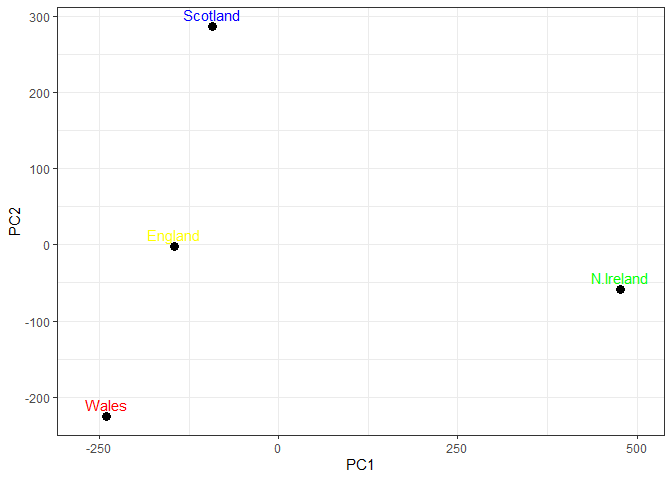
Q8. Customize your plot so that the colors of the country names match the colors in our UK and Ireland map and table at start of this document.
# Plot PC1 vs PC2 with ggplot
ggplot(pca$x) +
aes(x = PC1, y = PC2, label = rownames(pca$x)) +
geom_point(size = 3) +
geom_text(vjust = -0.5, col=c("yellow", "red", "blue", "green")) +
xlim(-270, 500) +
xlab("PC1") +
ylab("PC2") +
theme_bw()
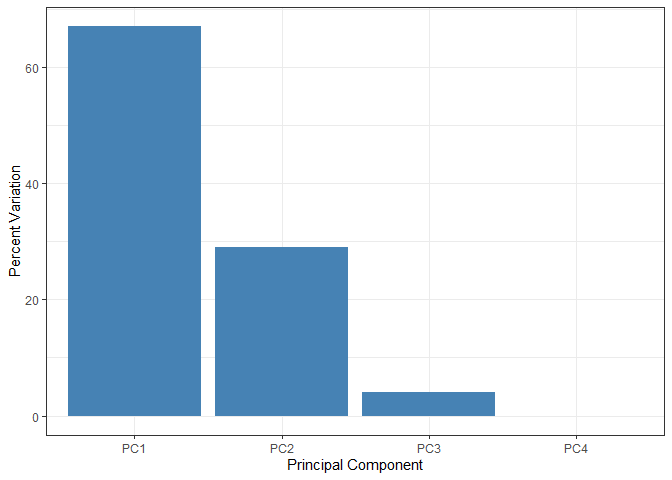
This calculation accoutns for how much of the variation in the data each PC accounts for
v <- round( pca$sdev^2/sum(pca$sdev^2) * 100 )
v
## [1] 67 29 4 0
## or the second row here...
z <- summary(pca)
z$importance
## PC1 PC2 PC3 PC4
## Standard deviation 324.15019 212.74780 73.87622 3.175833e-14
## Proportion of Variance 0.67444 0.29052 0.03503 0.000000e+00
## Cumulative Proportion 0.67444 0.96497 1.00000 1.000000e+00
# Create scree plot with ggplot
variance_df <- data.frame(
PC = factor(paste0("PC", 1:length(v)), levels = paste0("PC", 1:length(v))),
Variance = v
)
ggplot(variance_df) +
aes(x = PC, y = Variance) +
geom_col(fill = "steelblue") +
xlab("Principal Component") +
ylab("Percent Variation") +
theme_bw() +
theme(axis.text.x = element_text(angle = 0))
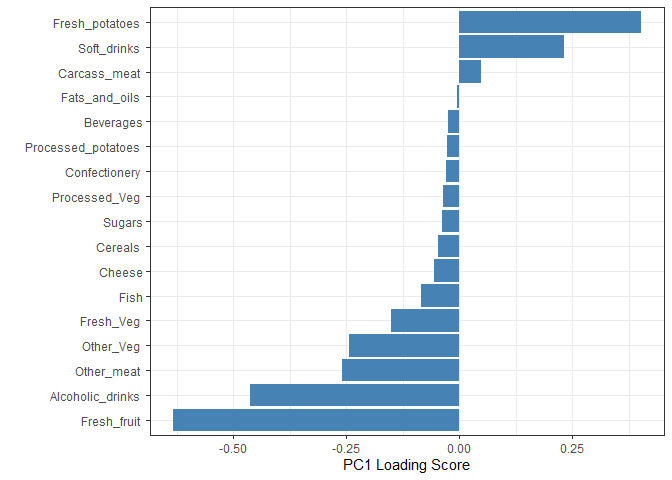
 Another major
result of PCA is the so-called “variable loadings” or
Another major
result of PCA is the so-called “variable loadings” or $rotation that
tells us how each of the original variables (i.e., foods) contributes to
our new axes (i.e., the PCs).
## Lets focus on PC1 as it accounts for > 90% of variance
ggplot(pca$rotation) +
aes(x = PC1,
y = reorder(rownames(pca$rotation), PC1)) +
geom_col(fill = "steelblue") +
xlab("PC1 Loading Score") +
ylab("") +
theme_bw() +
theme(axis.text.y = element_text(size = 9))
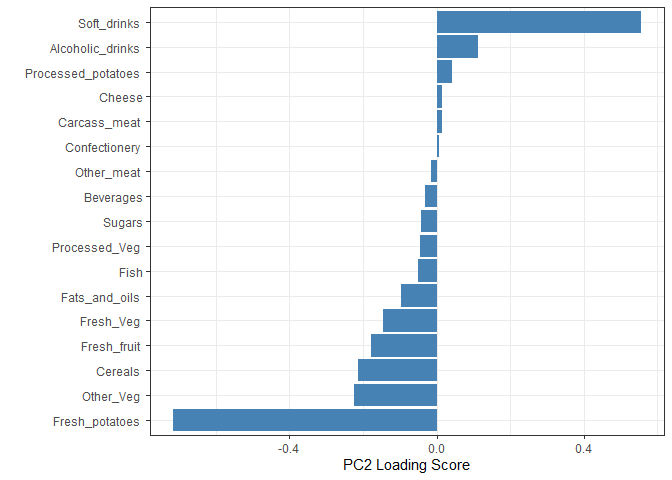
Q9. Generate a similar ‘loadings plot’ for PC2. What two food groups feature prominantely and what does PC2 mainly tell us about?
Soft drinks and fresh potatoes feature most prominently in this loading plot. It tells us which variables are driving the most variation away from the best fit line described by PC1 within each cluster.
ggplot(pca$rotation) +
aes(x = PC2,
y = reorder(rownames(pca$rotation), PC2)) +
geom_col(fill = "steelblue") +
xlab("PC2 Loading Score") +
ylab("") +
theme_bw() +
theme(axis.text.y = element_text(size = 9))